
Rugby Sodium Chloride Hypertonicity Ointment Recall?
Table of Contents
- How Can I Confirm if my Rugby Eye Drops Have Been Recalled?
- Where Can I Find the NDC Number on Rugby Sodium Chloride Hypertonicity Ointment and Solution?
- Is it possible Rugby Sodium Chloride Ointment and Solution Will be Recalled?
- What is Rugby Laboratories Saying About the Recall?
- Which Rugby Laboratories Eye Drops Were Recalled?
- Symptoms of Eye Infections
- What to Do if You Have Recalled Eye Drops
- Rugby Sodium Chloride Hypertonicity Ointment Recall: The Bottom Line
- Products Mentioned in this Blog Post
- About xeteor.com
- Full List of FDA Recalled Products: November 1st, 2023
Has Rugby Sodium Chloride Hypertonicity Ointment 5% Been Recalled?
The FDA recently recalled several over-the-counter eye drops sold at major retailers due to the potential risk of eye infections caused by bacterial contamination. A couple of Rugby Laboratories eye drops were included in this recall, so there have been many questions about exactly which eye drops are included in this recall. Learn if a Rugby Sodium Chloride Hypertonicity Ointment recall is imminent.
Many Rugby Sodium Chloride Hypertonicity Ointment 5% and Rugby Sodium Chloride Hypertonicity Ophthalmic Solution 5% customers have asked us about the recall, so we'll tell you specifically which Rugby eye drops have been recalled in this blog post. The recall is ongoing, so we recommend you visit the FDA website for the latest information.
There are currently NO warnings issued by the FDA for these two products:
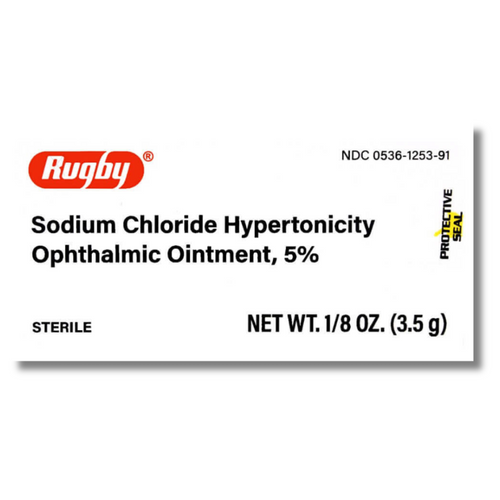
- Rugby Sodium Chloride Hypertonicity Ointment 5% is NOT included in the recall.
- Rugby Sodium Chloride Hypertonicity Solution 5% is NOT included in the recall.
How Can I Confirm if my Rugby Eye Drops Have Been Recalled?
Currently neither Rugby Sodium Chloride Hypertonicity Ointment nor Solution has been recalled. The best way to check is by comparing the NDC number on your product with the NDC numbers listed in the FDA Recall. NDC stands for National Drug Code and the number is unique for each product. Like a UPC code, the NDC number helps identify both over-the-counter and prescription products and they are especially useful in recall situations as it's easy to pinpoint EXACTLY which is product is safe to use and which product is recalled.
Where Can I Find the NDC Number on Rugby Sodium Chloride Hypertonicity Ointment and Solution?
1. Rugby Sodium Chloride Hypertonicity Ointment 5%
- NDC: 0536-1253-91 (circled in red)
- UPC: 305361253912
2. Rugby Sodium Chloride Hypertonicity Ophthalmic Solution 5%
- NDC: 0536-1254-94 (circled in red)
- UPC: 305361254940
Is it possible Rugby Sodium Chloride Ointment and Solution Will be Recalled?
While it is possible, it is unlikely as the recalled products are both linked to a 3rd party manufacturing plant owned by Velocity Pharma in India. Both Rugby Sodium Chloride Ointment and Solution are manufactured by Bausch & Lomb the maker of the brand name Muro 128, therefore they are unlikely to be recalled. We encourage you to visit the FDA website to get the latest information. A full list of Rugby and other brand name eye drops recalled by the FDA is here.
What is Rugby Laboratories Saying About the Recall?
As an authorized reseller of Rugby products, we contacted Rugby Laboratories directly to get more information. They said the recall is voluntary as no adverse reactions have been reported, however, this could change.
They post recall information in their news section (right side). Please note that Major Pharmaceuticals and Rugby Laboratories are the same company. As of writing this blog post, there was no further information regarding the eye drops in question.
If you would like to contact Rugby Laboratories Product Information Team directly, please call 1-800-616-2471 and select option 1. They are available Monday - Friday 8 am to 8 pm EST.
Currently there are no other recalls for Rugby Laboratories products, however, this is subject to change. We encourage you to visit the FDA website for the latest information.
Which Rugby Laboratories Eye Drops Were Recalled?
Below are the two Rugby eye drops with NDC and UPC codes that have been recalled by the FDA. Please check the NDC Number and UPC Code on the box to confirm products.
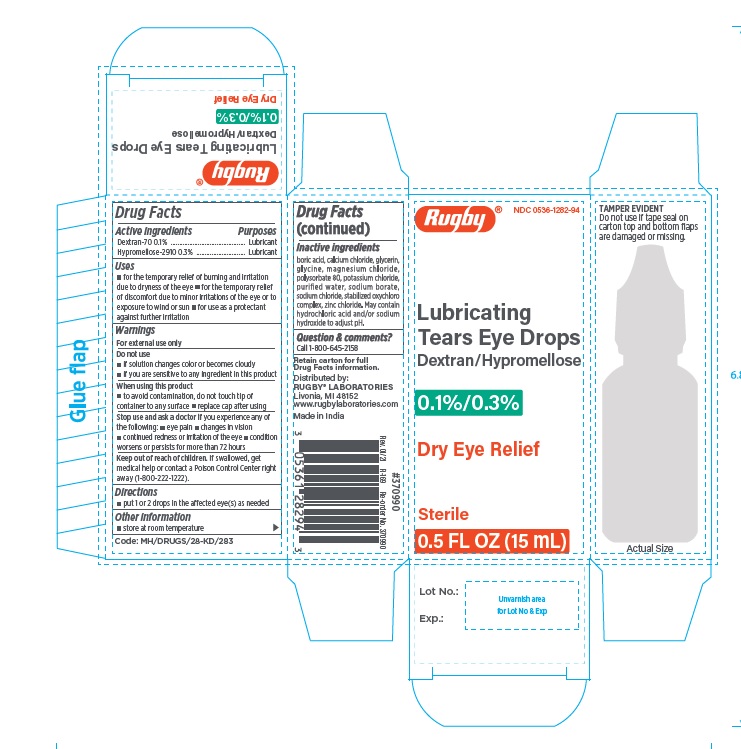
1. Rugby Lubricating Tears Eye Drops 0.1% Dextran / 0.3% Hypromellose 15 mL
- NDC Number: 0536-1282-94
- UPC Code: 305361282943
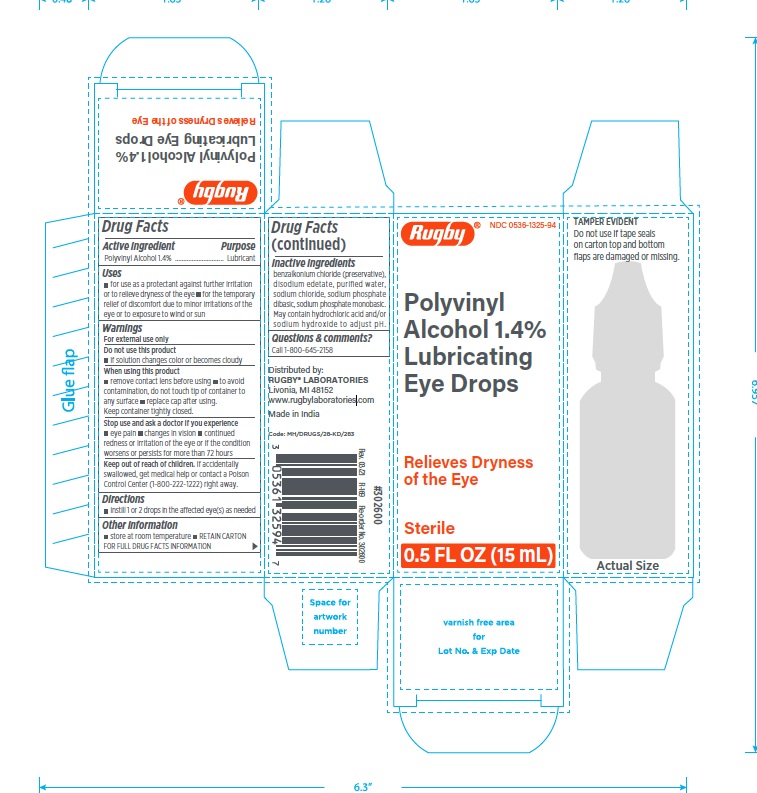
2. Rugby Polyvinyl Alcohol 1.4% Lubricating Eye Drops 15 mL
- NDC Number: 0536-1325-94
- UPC Code: 305361325947
Symptoms of Eye Infections
Using contaminated eye drops can lead to eye infections with symptoms like redness, swelling, eye pain, blurred vision, discharge, and swollen eyelids or conjunctiva. If you experience any of these symptoms after using the recalled drops, see a doctor immediately.
What to Do if You Have Recalled Eye Drops
If you have any of the recalled eye drops in your home, stop using them right away. Discard them safely by sealing the bottles in a plastic bag and throwing them in the trash. Never flush eye drops down the drain.
It's also a good idea to check your medicine cabinet for the affected products if you've purchased over-the-counter eye drops recently.
FDA has not received any adverse event reports of eye infection associated with these products at this time. FDA encourages health care professionals and patients to report adverse events or quality problems with any medicine to FDA’s MedWatch Adverse Event Reporting program:
- Complete and submit the report online at Medwatch
- Download and complete the form, then submit it via fax at 1-800-FDA-0178.
Rugby Sodium Chloride Hypertonicity Ointment Recall: The Bottom Line
Neither Rugby Sodium Chloride Hypertonicity Ointment 5% nor Rugby Sodium Chloride Hypertonicity Ophthalmic Solution 5% have been recalled, however, it's important to check both the FDA and Major Pharmaceuticals websites for the latest information. You can also contact Rugby Laboratories directly at 1-800-616-2471 and select option 1. They are available Monday - Friday 8 am to 8 pm EST.
Products Mentioned in this Blog Post
Rugby Sodium Chloride Hypertonicity Ophthalmic Ointment 5% | Muro 128
$14.99
Rugby Sodium Chloride Ophthalmic Ointment 5% – 3.5g Tube | Compare to Muro 128 Rugby Sodium Chloride Hypertonicity Ophthalmic Ointment 5% is a sterile, preservative-free...… read more
Rugby Sodium Chloride Hypertonicity Ointment 5% | Muro 128 (6 Pack)

$69.96
Rugby Sodium Chloride Ophthalmic Ointment 5% – 6 Pack (3.5g Tubes) | Generic for Muro 128 If you rely on sodium chloride ointment regularly, this...… read more
Rugby Sodium Chloride Hypertonicity Ophthalmic Solution 5% | Muro 128

$9.99
Rugby Sodium Chloride Ophthalmic Solution 5% – 0.5 fl oz Bottle | Compare to Muro 128 Rugby Sodium Chloride Hypertonicity Ophthalmic Solution 5% is a...… read more
Rugby Sodium Chloride Ophthalmic Solution 5% | Muro 128 (3 Pack)

$18.99
Rugby Sodium Chloride Ophthalmic Solution 5% – 3 Pack | Compare to Muro 128 Eye Drops Rugby Sodium Chloride Hypertonicity Ophthalmic Solution 5% provides fast,...… read more
About xeteor.com
xeteor.com, started in 2018 is based in Las Vegas, Nevada and is online pharmacy focusing on discount over-the-counter generic medications and diabetic test strips. We find deals normally reserved for "big pharma" and offer them to the consumer at discount prices. Ranking as the #1 authorized online reseller of Rugby Laboratories products according to Trustpilot.com, xeteor.com has 8,000+ 5-star reviews online, including Amazon, eBay, Trustpilot, and Google. xeteor.com is a Google Top Quality Store, earned by merchants that provide a great shopping experience.
Shipping Time & Cost
All items ship same day when purchased by 3 pm PST Monday - Friday. We offer free shipping nationwide with no minimum order. Shipping times are between 3 to 8 business days, with an average of 3.3 business days. A link to our full shipping policy is here.
Payment Options
We offer multiple convenient payment options, including Visa, Mastercard, Discover, American Express, Google Pay, Apple Pay, PayPal and Venmo.
Return Policy
We offer a 100% satisfaction guarantee. If you are not satisfied with the product, you may return it up to 100 days from the date of purchase. We offer free return shipping to ensure you can try the product risk free. A link to our full return policy is here.
How to Reach Us?
If you have questions about the product or need assistance, we are happy to talk on the phone. Call us at 1-833-787-4777 between 9 am - 5 pm PST Monday - Friday. You can also email us at [email protected] or text us at 702-659-7022. A link to our full contact us page is here.
Disclaimer: This post is intended for informational purposes only. It is not a substitute for professional medical advice, diagnosis, or treatment. Always consult your doctor or another qualified health provider with any questions you may have regarding a medical condition or before starting any new treatment. Do not disregard professional medical advice or delay in seeking it because of something you have read in this post.
Full List of FDA Recalled Products: November 1st, 2023
Please visit the FDA website for the latest list.
| Retailer/Label | Product | Product Information |
|---|---|---|
| CVS Health | Lubricant Eye Drops 15 ml (single pack) | Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
| CVS Health | Lubricant Eye Drops 15 ml (twin pack) | Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
| CVS Health | Lubricant Gel Drops 15 ml (single pack) | Carboxymethylcellulose Sodium Eye Drops 1% w/v |
| CVS Health | Lubricant Gel Drops 15 ml (twin pack) | Carboxymethylcellulose Sodium Eye Drops 1% w/v |
| CVS Health | Multi-Action Relief Drops 15 ml | Polyvinyl Alcohol 0.5% w/v & Povidone 0.6% w/v & Tetrahydrozoline Hydrochloride 0.05% Eye Drops |
| CVS Health | Lubricating Gel drops 10 ml | Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
| CVS Health | Lubricant Eye Drops 10 ml (single pack) | Propylene Glycol Eye Drops 0.6% w/v |
| CVS Health | Lubricant Eye Drops 10 ml (twin pack) | Propylene Glycol Eye Drops 0.6% w/v |
| CVS Health | Mild Moderate Lubricating Eye Drops 15 ml (single pack) | Polyethylene Glycol 400 Eye Drop '0.25% w/v |
| Rugby (Cardinal Health) | Lubricating Tears Eye Drops 15 ml | Hypromellose 2910-0.3% w/v & Dextran 70- 0.1% Eye Drops |
| Rugby (Cardinal Health) | Polyvinyl Alcohol 1.4% Lubricating Eye Drops 15 ml | Polyvinyl Alcohol Eye Drops 1.4% w/v |
| Leader (Cardinal Health) | Dry Eye Relief 10 ml | Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
| Leader (Cardinal Health) | Lubricant Eye Drops 15 ml (single pack) | Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
| Leader (Cardinal Health) | Lubricant Eye Drops 15 ml (twin pack) | Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
| Leader (Cardinal Health) | Dry Eye Relief 15 ml | Carboxymethylcellulose Sodium Eye Drops 1% w/v |
| Leader (Cardinal Health) | Eye Irritation Relief 15 ml | Polyvinyl Alcohol 0.5% w/v & Povidone 0.6% w/v & Tetrahydrozoline Hydrochloride 0.05% Eye Drops |
| Rite Aid | Lubricant Eye Drops 15 ml (twin pack) | Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
| Rite Aid | Lubricant Eye Drops 10 ml (twin pack) | Propylene Glycol Eye Drops 0.6% w/v |
| Rite Aid | Gentle Lubricant Gel Eye Drops 15 ml | Hypromellose 0.3%, Glycerin 0.2%, Dextran 70 0.1% Eye Drops |
| Rite Aid | Lubricant Gel Drops 15 ml | Carboxymethylcellulose Sodium Eye Drops 1% w/v |
| Rite Aid | Lubricating Gel Drops 10 ml | Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
| Rite Aid | Multi-Action Relief Drops 15 ml | Polyvinyl Alcohol 0.5% w/v & Povidone 0.6% w/v & Tetrahydrozoline Hydrochloride 0.05% Eye Drops |
| Target | Up&Up Dry Eye Relief Lubricant Eye Drops 30 ml | Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
| Target | Up&Up Extreme Relief Dry Eye 15 ml (single pack) | Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |
| Target | Up&Up Extreme Relief Dry Eye 30 ml (twin pack) | Carboxymethylcellulose Sodium Eye Drops 0.5% w/v |
| Velocity Pharma LLC | Lubricant Eye Drop 10 ml (triple pack) | Propylene Glycol Eye Drops 0.6% w/v |
| Walmart | Equate Hydration PF Lubricant Eye Drop 10 ml | Polyethylene Glycol 400 0.4% & Propylene Glycol 0.3% Eye Drops |






